Novel metal free porphyrinic photosensitizers supported on solvent-induced Amberlyst-15 nanoparticles with a porous structure†
Abstract
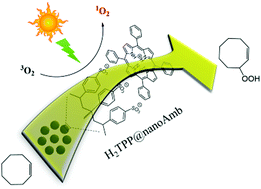
In this study, a series of meso-tetraarylporphyrins (aryl = phenyl, 2-methylphenyl and 2-chlorophenyl), H2T(aryl)P immobilized on nanosized Amberlyst-15 (nanoAmb) through the acid–base reaction were utilized as highly efficient (turnover number in the range of 4 × (8000 to 10 000) for 4 successive reactions with the same catalyst), stable and reusable heterogeneous photocatalysts for the aerobic oxidation of olefins under green conditions. Interestingly, nanoAmb with an average diameter of less than 200 nm was produced from Amb beads by overnight magnetic stirring in ethyl acetate at room temperature. The catalysts may be reused at least four times without a significant decrease in catalytic activity. The immobilization of porphyrin on the polymer was confirmed by diffuse reflectance UV-vis and IR spectroscopy. Also, the supported porphyrin was released from the nanoAmb surface by alkaline treatment and analyzed by UV-vis spectroscopy. N2 porosimetry analysis showed a significant decrease in the BET surface area and pore volume values of the polymer after the immobilization of porphyrins. The influence of different parameters, including the photosensitizer to olefin molar ratio, porphyrin loading, solvent, type of light source and meso substituents were investigated. H2T(2-Me)PP@nanoAmb and H2T(2-Cl)PP@nanoAmb showed the highest oxidative stability and catalytic activity among the series of porphyrins, respectively. In order to overcome the photooxidative degradation of porphyrin photosensitizers, different degrees of catalyst loading were examined and the lowest loading showed the best performance. The use of 1,4-benzoquinone and 1,3-diphenylisobenzofuran as the quenchers of the superoxide anion radical and singlet oxygen species revealed that the oxidation reaction mainly proceeds with a singlet oxygen mechanism.


 Please wait while we load your content...
Please wait while we load your content...