Synthesis and characterization of low viscosity hexafluoroacetylacetonate-based hydrophobic magnetic ionic liquids†
Abstract
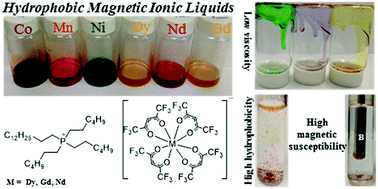
Magnetic ionic liquids (MILs) are distinguished from traditional ionic liquids (ILs) by the incorporation of a paramagnetic component within their chemical structure. Hydrophobic MILs are novel solvents that can be used in many applications, including liquid–liquid extraction (LLE) and catalysis. Low viscosity and low water solubility are essential features that determine their feasibility in LLE. In this study, extremely hydrophobic MILs were synthesized by using transition and rare earth metal hexafluoroacetylacetonate chelated anions paired with the trihexyl(tetradecyl)phosphonium ([P66614+]) cation. Hydrophobic MILs exhibiting water solubilities less than 0.01% (v/v) were synthesized in a rapid two-step procedure. Furthermore, the viscosities of the MILs are among some of the lowest ever reported for hydrophobic MILs (276.5–927.9 centipoise (cP) at 23.7 °C) dramatically improving the ease of handling these liquids. For the first time, the magnetic properties of MILs possessing hexafluoroacetylacetonate chelated metal anions synthesized in this study are reported using a superconducting quantum interference device (SQUID) magnetometer. Effective magnetic moments (μeff) as high as 9.7 and 7.7 Bohr magnetons (μB) were achieved by incorporating high spin dysprosium and gadolinium ions, respectively, into the anion component of the MIL. The low viscosity, high hydrophobicity, and large magnetic susceptibility of these MILs make them highly attractive and promising solvents for separations and purification, liquid electrochromic materials, catalytic studies, as well as microfluidic applications.


 Please wait while we load your content...
Please wait while we load your content...