Synthesis of thioether andrographolide derivatives and their inhibitory effect against cancer cells†‡
Abstract
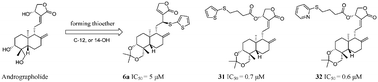
A series of novel thioether andrographolide derivatives were synthesized by incorporating various aromatic (or heteroaromatic) substituents into C-12 or 14-OH. A total of 38 andrographolide derivatives were prepared and evaluated for their in vitro inhibitory activity against cancer cells. All the derivatives exhibited better activity against prostate cancer cells (PC-3) than the parent compound. Among these, compounds 6a, 8, 9, 17, 19, 31, and 32 demonstrated good activity. These compounds were further evaluated for their anticancer activities against other cancer cell lines including MCF-7, MDA-MB-231, and A549. Compounds 31 and 32 showed excellent activity against MCF-7 with an IC50 value of 0.7 and 0.6 μM, respectively. The absolute configuration of 15a was determined via single-crystal X-ray diffraction. The activity of 6a (12S), which was the precursor of 15a, was better than that of the diastereoisomer 6b (12R). Moreover, the preliminary structure–activity relationship has been summarized. The results obtained herein are very important for further optimization of andrographolide.


 Please wait while we load your content...
Please wait while we load your content...