A highly-occupied, single-cell trapping microarray for determination of cell membrane permeability
Abstract
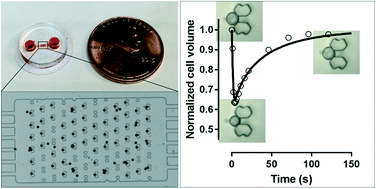
Semi- and selective permeability is a fundamentally important characteristic of the cell membrane. Membrane permeability can be determined by monitoring the volumetric change of cells following exposure to a non-isotonic environment. For this purpose, several microfluidic perfusion chambers have been developed recently. However, these devices only allow the observation of one single cell or a group of cells that may interact with one another in an uncontrolled way. Some of these devices have integrated on-chip temperature control to investigate the temperature-dependence of membrane permeability, but they inevitably require sophisticated fabrication and assembly, and delicate temperature and pressure calibration. Therefore, it is highly desirable to design a simple single-cell trapping device that allows parallel monitoring of multiple separate, individual cells subjected to non-isotonic exposure at various temperatures. In this study, we developed a pumpless, single-layer microarray with high trap occupancy of single cells. The benchmark performance of the device was conducted by targeting spherical particles of 18.8 μm in diameter as a model, yielding trap occupancy of up to 86.8% with a row-to-row shift of 10–30 μm. It was also revealed that in each array the particles larger than a corresponding critical size would be excluded by the traps in a deterministic lateral displacement mode. Demonstrating the utility of this approach, we used the single-cell trapping device to determine the membrane permeability of rat hepatocytes and patient-derived circulating tumor cells (Brx-142) at 4, 22 and 37 °C. The membrane of rat hepatocytes was found to be highly permeable to water and small molecules such as DMSO and glycerol, via both lipid- and aquaporin-mediated pathways. Brx-142 cells, however, displayed lower membrane permeability than rat hepatocytes, which was associated with strong coupling of water and DMSO transport but less interaction between water and glycerol. The membrane permeability data reported here provide new insights into the biophysics of membrane transport such as aquaporin expression and coupling transport of water and solutes, as well as providing essential data for the ultimate goal of biobanking rare cells and precious tissues.


 Please wait while we load your content...
Please wait while we load your content...