Whole-cell biocatalytic selective oxidation of 5-hydroxymethylfurfural to 5-hydroxymethyl-2-furancarboxylic acid†
Abstract
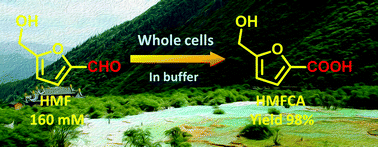
Currently, the catalytic upgradation of 5-hydroxymethylfurfural (HMF), an important bio-based platform chemical, is of great interest. In this work, we reported the biocatalytic oxidation of HMF to 5-hydroxymethyl-2-furancarboxylic acid (HMFCA) by using a newly isolated strain Comamonas testosteroni SC1588. Compared to growing cells, resting cells were found to be a better biocatalyst for the synthesis of HMFCA. This strain exhibited high tolerance to HMF (up to 180 mM). However, its catalytic performances were highly sensitive to pH. The product exerted a significant negative effect on the catalytic performances and viability of the cells, partially due to the acidity of this compound. The product inhibition and toxicity toward this strain were reduced considerably after adjusting the pH of the reaction mixtures to 7.0. Excellent substrate conversions (approximately 100%) and good HMFCA yields (88–99%) were obtained when the substrate concentrations were less than 130 mM. In addition, the HMFCA synthesis could be improved significantly by the combination of histidine addition with pH tuning at higher substrate concentrations. Catalytic activities of the cells increased markedly when they were cultivated in the presence of a low concentration of furfural and furfuryl alcohol. HMFCA was afforded in a yield of approximately 98% after 36 h at a substrate concentration of up to 160 mM. Besides, this strain was capable of selectively transforming other furfurals to the target acids with good yields of 90–93%.
- This article is part of the themed collection: International Symposium on Green Chemistry 2017


 Please wait while we load your content...
Please wait while we load your content...