Joint tuning of nanostructured Cu-oxide morphology and local electrolyte programs high-rate CO2 reduction to C2H4†
Abstract
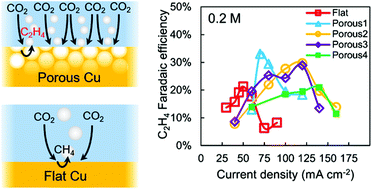
Electrochemical ethylene production rates are enhanced by pushing favourable local electrolyte conditions to occur at higher current densities and lower relative overpotentials. In particular the combined influences of electrode morphology and buffering on electrode pH and CO2 conditions are assessed.


 Please wait while we load your content...
Please wait while we load your content...