Selective conversion of bio-derived ethanol to renewable BTX over Ga-ZSM-5†
Abstract
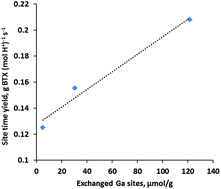
Selective conversion of bio-derived ethanol to benzene, toluene and xylenes (BTX) is desirable for producing renewable BTX. In this work, we show that addition of Ga to H-ZSM-5 leads to a two-fold increase in the BTX yield as compared with H-ZSM-5 when ethanol is converted over these zeolites at 450 °C and ambient pressure. Besides promoting BTX formation, Ga also plays an important role in enhancing molecular hydrogen production and suppressing hydrogen transfer reactions for light alkane formation. The ion exchange synthesis of Ga-ZSM-5 results in the majority of Ga at the outer surface of zeolite crystals as extra-zeolitic Ga2O3 particles and only a small fraction of Ga exchanging with the Brønsted acid sites which appears to be responsible for higher ethanol conversion to BTX. The interface between H-ZSM-5 and Ga2O3 particles is not active since H-ZSM-5 and the physical mixture of β-Ga2O3/H-ZSM-5 furnish an almost identical product distribution. Hydrogen reduction of the physical mixtures facilitates movement of Ga to ion exchange locations and dramatically increases the BTX yield becoming comparable to those obtained over ion-exchanged Ga-ZSM-5, suggesting that exchanged Ga(III) cations are responsible for the increased BTX production. A linear correlation between the BTX site time yield and exchanged Ga sites further confirms that Ga occupying cationic sites are active sites for enhancing BTX formation. Reduction of physical mixtures (β-Ga2O3/H-ZSM-5) also provides an economical and environmentally friendly non-aqueous method for large scale catalyst synthesis without sacrificing catalyst performance for ethanol conversion application.
- This article is part of the themed collection: 2017 Green Chemistry Hot Articles


 Please wait while we load your content...
Please wait while we load your content...