Informing rational design of graphene oxide through surface chemistry manipulations: properties governing electrochemical and biological activities†
Abstract
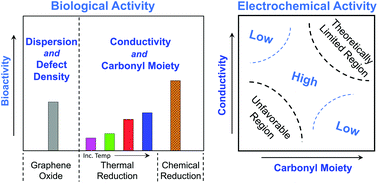
It is increasingly realized that rational design is critical to advance potential applications and proactively preclude adverse consequences of carbon nanomaterials (CNMs). Central to this approach is the establishment of parametric relationships that correlate material properties to both their functional performance and inherent hazard. This work aims to decouple the causative mechanisms of material structure and surface chemistry as it relates to the electrochemical and biological activities of graphene oxide (GO). The results are evaluated in the context of established relationships between surface chemistry and oxygen functionalized multi-walled carbon nanotubes (O-MWCNTs), a carbon allotrope. Systematic manipulation of GO surface chemistry is achieved through thermal annealing (under inert conditions, 200–900 °C). To further elucidate the contribution of several properties, chemical reduction was also used as an approach to differentially modify the surface chemistry. Physicochemical properties of GO and reduced GO (rGO) samples were comprehensively characterized using multiple techniques (AFM, TGA, XPS, ATR-FTIR, Raman, and DLS). The results indicate that surface chemistry is a viable design handle to control both activities. Rather than a single direct property (i.e., relative presence of carbonyl-containing moieties), it is a balance of multiple consequential properties, (extent of dispersion, defect density, and electrical conductivity) in combination with the relative presence of carbonyl moieties that synergistically contribute to electrochemical and biological activities. The identification of these governing physicochemical properties aims to inform the establishment of design parameters to guide the rational and safe design of CNMs.
- This article is part of the themed collection: Green Chemistry 2017 Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...