Preparation, characterization and bioavailability by oral administration of O/W curcumin nanoemulsions stabilized with lysophosphatidylcholine
Abstract
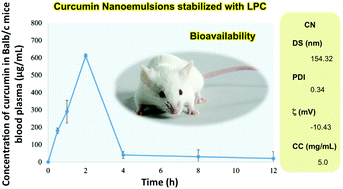
Curcumin is the main and most abundant bioactive component in Curcuma longa L. with documented properties in the prevention and treatment of chronic degenerative and infectious diseases. However, curcumin has low solubility in aqueous media, hence low bioavailability when administered orally. The use of nanoemulsions as carriers can provide a partial solution to bioavailability restrictions. In our study, O/W nanoemulsions of curcumin were prepared using lysophosphatidylcholine, a phospholipid with proven emulsification capacity; nevertheless, such qualities have not been previously reported in the preparation of nanoemulsions. Lysophosphatidylcholine was obtained by enzymatic removal of one fatty acid residue from phosphatidylcholine. The objective of our work was to formulate stable curcumin nanoemulsions and evaluate their bioavailability in BALB/c mice plasma after oral administration. Formulated nanoemulsions had a droplet size mean of 154.32 ± 3.10 nm, a polydispersity index of 0.34 ± 0.07 and zeta potential of −10.43 ± 1.10 mV; stability was monitored for 12 weeks. Lastly, in vivo pharmacokinetic parameters, using BALB/c mice, were obtained; namely, Cmax of 610 ± 65.0 μg mL−1 and Tmax of 2 h. Pharmacokinetic data revealed a higher bioavailability of emulsified as opposed to free curcumin. Research regarding other potential emulsifiers that may provide better health benefits and carry nano-encapsulated bioactive compounds more effectively, is necessary. This study provides important data on the preparation and design of nanoencapsulated Curcumin using lysophosphatidylcholine as an emulsifier.


 Please wait while we load your content...
Please wait while we load your content...