Sulfidation kinetics of copper oxide nanoparticles†
Abstract
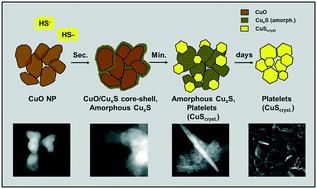
Sulfidation of copper oxide nanoparticles (CuO NPs) in urban wastewater systems is expected to influence their impact on the environment. However, the kinetics of this reaction has not been studied to date and the reaction mechanism remains largely unexplored. We therefore investigated the sulfidation kinetics of CuO NPs reacted with bisulfide (HS−) at concentrations relevant to wastewater systems. Pristine CuO NPs (50 nm, 7.7 μM) were reacted with HS− (26.4–105.6 μM) in oxic solutions buffered to pH 8.0. The reaction progress was monitored using silver nitrate to quench the reaction and selectively dissolve the copper sulfides (CuxS) and zincon to spectrophotometrically quantify the released Cu2+. In addition, the reaction products were characterized at selected time points using analytical electron microscopy and X-ray absorption spectroscopy (XAS). The sulfidation rate of the CuO NPs was best described by a pseudo first order rate law and the corresponding half-life times ranged between 1 and 6 minutes. XAS results showed that crystalline CuO NPs rapidly transformed into amorphous CuxS and gradually into crystalline CuS (covellite). The comparable size of pristine and transformed primary particles, the similar morphology of their aggregates, and the initial formation of CuO–CuxS core–shell structures revealed by analytical electron microscopy suggest that the initial sulfidation occurred via a direct conversion reaction mechanism. Our findings suggest that CuO NPs released from various sources into wastewater will rapidly transform into amorphous CuxS and eventually recrystallize into covellite.


 Please wait while we load your content...
Please wait while we load your content...