Transformations of silver nanoparticles in wastewater effluents: links to Ag bioavailability†
Abstract
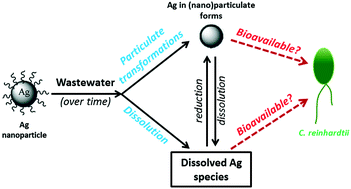
Wastewater effluents represent one of the main routes through which silver nanoparticles (Ag NPs) can reach natural aquatic environments, where they may interact with a range of organisms. However, in wastewaters, Ag NPs may undergo different chemical and physical transformations, which may alter NP toxicity towards aquatic organisms. The main objectives of this study are to characterize the dissolution and speciation of a Ag NP in a wastewater effluent and to assess its interactions with a model organism, Chlamydomonas reinhardtii (green alga), in the same medium. Experiments were conducted to distinguish the effect(s) of the wastewater matrix on the dissolution of Ag NPs and to determine whether the transformed NPs or the dissolved Ag species would be more bioavailable to C. reinhardtii. It was shown that in the wastewater effluent, Ag bioavailability was significantly reduced with respect to experiments using similar Ag concentrations in simple media. The substantial reduction in Ag bioavailability in wastewaters could be explained by the presence of high concentrations of organic/inorganic ligands, which complexed the Ag and the presence of substantial competing ions. Nonetheless, Ag NPs did appear to be bioavailable to C. reinhardtii at higher concentrations, as observed from bioaccumulation experiments comparing dissolved Ag and Ag NPs. Since the biouptake of Ag NPs could not be explained by concentrations of free Ag alone, it was hypothesized that complexed Ag species and/or small NPs may have contributed to biouptake. The results suggested that the chemistry of wastewaters had a significant effect on the bioavailability of Ag NPs, by modifying their physicochemical transformations.
- This article is part of the themed collection: CSC100: Celebrating Canadian Chemistry


 Please wait while we load your content...
Please wait while we load your content...