Predictability of silver nanoparticle speciation and toxicity in ecotoxicological media†
Abstract
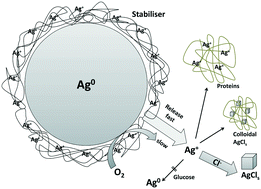
The use of silver nanoparticles (AgNPs) as an antimicrobial agent has increased significantly over the past decade which potentiates their release to the environment. The antimicrobial effect of AgNPs is generally considered to be due to the release of silver ions (Ag+). Here we describe their bioavailability under environmental conditions and demonstrate its influence on (eco-)toxicity for the AgNP NM-300K and a wide variety of aquatic organisms (green algae, plants and crustaceans), terrestrial organisms (soil bacteria) and mammalian cells. Since the bioavailability of AgNPs is largely determined by Ag speciation, this paper focuses on the Ag speciation in the test media for these organisms. We predicted the Ag speciation in aqueous test media by equilibrium speciation calculation and validated the results by comparison with experimental speciation using ultracentrifugation and membrane filtration. Silver amounts were quantified using GF-AAS, ICP-OES/MS and UV-vis. The dissolved Ag concentrations were controlled by the fast initial release of a limited amount of Ag+. After this initial release, the media components, chloride and proteins, controlled the available dissolved Ag by precipitation and complexation. Further release of Ag+ due to oxidation was not observed in the time frame of our experiments, except for media with very high chloride content. Apparently, the stabilisers of these AgNPs prevented any further release accounting for an enhanced redox stability. These findings facilitated the prediction of the bioavailability of Ag in the test media and, based on literature toxicity data, also of its toxic effects (EC50) on the respective organisms. The toxic effects of the AgNP NM-300K depended solely on the amount of Ag+ that was already present in the stock dispersion and not from further release due to later oxidation processes.


 Please wait while we load your content...
Please wait while we load your content...