Improving the accuracy of effect-directed analysis: the role of bioavailability
Abstract
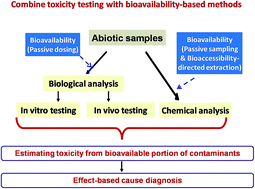
Aquatic ecosystems have been suffering from contamination by multiple stressors. Traditional chemical-based risk assessment usually fails to explain the toxicity contributions from contaminants that are not regularly monitored or that have an unknown identity. Diagnosing the causes of noted adverse outcomes in the environment is of great importance in ecological risk assessment and in this regard effect-directed analysis (EDA) has been designed to fulfill this purpose. The EDA approach is now increasingly used in aquatic risk assessment owing to its specialty in achieving effect-directed nontarget analysis; however, a lack of environmental relevance makes conventional EDA less favorable. In particular, ignoring the bioavailability in EDA may cause a biased and even erroneous identification of causative toxicants in a mixture. Taking bioavailability into consideration is therefore of great importance to improve the accuracy of EDA diagnosis. The present article reviews the current status and applications of EDA practices that incorporate bioavailability. The use of biological samples is the most obvious way to include bioavailability into EDA applications, but its development is limited due to the small sample size and lack of evidence for metabolizable compounds. Bioavailability/bioaccessibility-based extraction (bioaccessibility-directed and partitioning-based extraction) and passive-dosing techniques are recommended to be used to integrate bioavailability into EDA diagnosis in abiotic samples. Lastly, the future perspectives of expanding and standardizing the use of biological samples and bioavailability-based techniques in EDA are discussed.


 Please wait while we load your content...
Please wait while we load your content...