Emerging investigator series: As(v) in magnetite: incorporation and redistribution†
Abstract
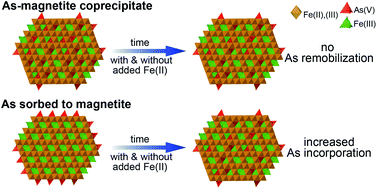
Exposure to As in groundwater negatively impacts millions of people around the globe, and As mobility in groundwater is often controlled by Fe mineral dissolution and precipitation. Additionally, trace elements can be released from and incorporated into the structure of Fe oxides in the presence of dissolved Fe(II). The potential for As to redistribute between sorbed on the magnetite surface and incorporated in the magnetite structure, however, remains unclear. In this study, we use selective chemical extraction and X-ray absorption spectroscopy (XAS) to distinguish magnetite-sorbed and incorporated As(V) and to provide evidence for As(V) incorporation during magnetite precipitation. While As in the As-magnetite coprecipitates did not redistribute between sorbed and incorporated over a 4 month period, a small, but measurable increase in incorporated As(V) of up to 13% was observed for sorbed As(V). We suggest that Fe(II)-catalyzed recrystallization of magnetite did not significantly influence the redistribution of sorbed As(V) because the extent of Fe atom exchange was small (∼10%). In addition, the extent of As redistribution was the same in the absence and presence of added aqueous Fe(II), suggesting that aqueous Fe(II) had, overall, a minor effect on As redistribution for both coprecipitated and sorbed As(V). Our results suggest that coprecipitation of As(V) with magnetite and redistribution of As(V) sorbed on magnetite are potential pathways for irreversible As(V) uptake and sequestration. These pathways are likely to play a significant role in controlling As mobility in natural systems, during human-induced redox cycling of groundwater such as aquifer storage and recovery, as well as in iron oxide-based As removal systems.
- This article is part of the themed collection: Emerging Investigator Series


 Please wait while we load your content...
Please wait while we load your content...