Isotope fractionation associated with the simultaneous biodegradation of multiple nitrophenol isomers by Pseudomonas putida B2
Abstract
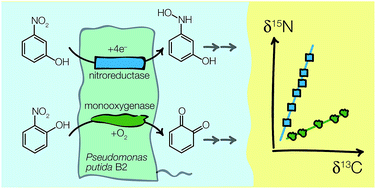
Quantifying the extent of biodegradation of nitroaromatic compounds (NACs) in contaminated soils and sediments is challenging because of competing oxidative and reductive reaction pathways. We have previously shown that the stable isotope fractionation of NACs reveals the routes of degradation even if it is simultaneously caused by different bacteria. However, it is unclear whether compound-specific isotope analysis (CSIA) can be applied in situations where multiple pollutants are biodegraded by only one microorganism under multi-substrate conditions. Here we examined the C and N isotope fractionation of 2-nitrophenol (2-NP) and 3-nitrophenol (3-NP) during biodegradation by Pseudomonas putida B2 through monooxygenation and partial reductive pathways, respectively, in the presence of single substrates vs. binary substrate mixtures. Laboratory experiments showed that the reduction of 3-NP by Pseudomonas putida B2 is associated with large N and minor C isotope fractionation with C and N isotope enrichment factors, εC and εN, of −0.3 ± 0.1‰ and −22 ± 0.2‰, respectively. The opposite isotope fractionation trends were found for 2-NP monooxygenation. In the simultaneous presence of 2-NP and 3-NP, 2-NP is biodegraded at identical rate constants and εC and εN values (−1.0 ± 0.1‰ and −1.3 ± 0.2‰) to those found for the monooxygenation of 2-NP in single substrate experiments. While the pathway and N isotope fractionation of 3-NP reduction (εN = −24 ± 1.1‰) are independent of the presence of 2-NP, intermediates of 2-NP monooxygenation interfere with 3-NP reduction. Because neither pH, substrate uptake, nor aromatic substituents affected the kinetic isotope effects of nitrophenol biodegradation, our study illustrates that CSIA provides robust scientific evidence for the assessment of natural attenuation processes.


 Please wait while we load your content...
Please wait while we load your content...