Concentrations of a triplet excited state are enhanced in illuminated ice†
Abstract
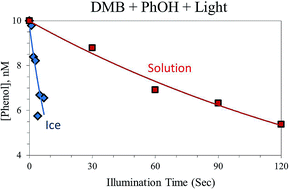
Photochemical reactions influence the fates and lifetimes of organic compounds in snow and ice, both through direct photoreactions and via photoproduced transient species such as hydroxyl radical (˙OH) and, perhaps, triplet excited states of organic compounds (i.e., triplets). While triplets can be important oxidants in atmospheric drops and surface waters, little is known of this class of oxidants in frozen samples. To investigate this, we examined the photoreaction of phenol with the triplet state of 3,4-dimethoxybenzaldehyde (3DMB*), a product from biomass combustion, in illuminated laboratory ices. Our results show that the rate of phenol loss due to 3DMB* is, on average, increased by a factor of 95 ± 50 in ice compared to the equivalent liquid sample. We find that this experimentally measured freeze concentration factor, FEXP, is independent of total solute concentration and temperature, in contrast to what is expected from a liquid-like region whose composition follows freezing point depression. We also find that FEXP for triplets is independent of pH, although the rates of phenol loss increase with decreasing pH in both solution and ice. The enhancement in the rate of phenol loss in/on ice indicates that concentrations of triplet excited states are enhanced in ice relative to solution and suggests that this class of oxidants might be a significant sink for organics in snow and ice.
- This article is part of the themed collection: Editor’s Choice: Aquatic Photochemistry


 Please wait while we load your content...
Please wait while we load your content...