Cu nanowires shelled with NiFe layered double hydroxide nanosheets as bifunctional electrocatalysts for overall water splitting†
Abstract
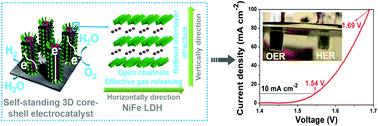
Developing highly active and low-cost electrocatalysts with superior durability for both the oxygen evolution reaction (OER) and hydrogen evolution reaction (HER) is a grand challenge to produce hydrogen by electrolysis of water. Here, we report on a facile and scalable approach to fabricate highly efficient three-dimensional (3D) bulk catalysts of core–shell nanostructures, in which few-layer NiFe layered double hydroxide (LDH) nanosheets are grown on Cu nanowire cores supported on Cu foams, toward overall water splitting. Remarkably, benefiting from the 3D hierarchical nanoarchitecture with large surface areas, fast electron transport, and open-channels for effective gas release, the resulting 3D self-standing catalysts exhibit outstanding OER activity as well as excellent HER performance in an alkaline medium. Using them as bifunctional catalysts for overall water splitting, a current density of 10 mA cm−2 was achieved at a voltage of 1.54 V, and 100 mA cm−2 at 1.69 V with excellent durability, which is much better than the benchmark of IrO2(+)//Pt(−) electrodes. Our 3D core–shell electrocatalysts significantly advance the research towards large-scale practical water electrolysis.
- This article is part of the themed collection: 2017 Energy and Environmental Science HOT articles


 Please wait while we load your content...
Please wait while we load your content...