Cooperative water oxidation catalysis in a series of trinuclear metallosupramolecular ruthenium macrocycles†
Abstract
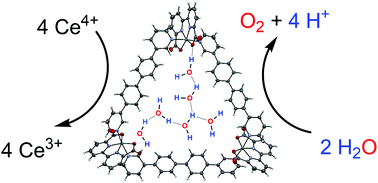
A series of trinuclear metallosupramolecular Ru(bda) macrocycles has been synthesized and the impact of the ring size on the catalytic activity in homogeneous water oxidation has been elucidated (bda = 2,2′-bipyridine-6,6′-dicarboxylate). Kinetic experiments revealed that for all macrocyclic catalysts a proton-coupled oxidation event is rate-determining. Distinct H/D kinetic isotope effects (KIE) have been observed for the individual macrocycles, which follow the same tendency like the catalytic activity where an intermediate ring size exhibits the best performance. To get insight into the different molecular properties, molecular dynamics simulations using a QM/MM approach have been performed for all reaction steps, revealing that the size of the cavity strongly influences the hydrogen-bonded water network inside the macrocyclic structure and thus, important proton-coupled reaction steps of the catalytic cycle. For the most active catalysts, all reactive sites point into the interior of the cavity, which is supported by a single crystal X-ray analysis of the largest macrocycle, and a cooperative effect between the individual Ru(bda) centers facilitating proton abstraction has been substantiated. On the other hand, different structural conformations predominate the smaller macrocycles, which prevent efficient interactions between the Ru(bda) centers.
- This article is part of the themed collection: 2017 Energy and Environmental Science HOT articles


 Please wait while we load your content...
Please wait while we load your content...