Ultra-high cycling stability of poly(vinylphenothiazine) as a battery cathode material resulting from π–π interactions†
Abstract
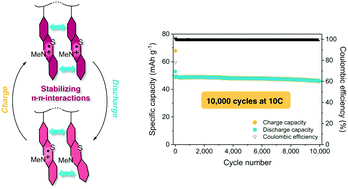
Organic cathode materials are promising candidates for a new generation of “green batteries”, since they have low toxicity and can be produced from renewable resources or from oil. Especially suitable are organic redox polymers that can be reversibly oxidized and reduced. Because of their often-insulating nature, however, many redox polymers have limited rate capabilities. Their cycling stabilities, which are of high importance for the long cycle-life of a battery cell, rarely exceed 1000 cycles. Here, we present a new concept for redox polymers as cathode materials, in which the oxidized states are stabilized through π–π interactions between redox-active groups. We found that due to these interactions poly(3-vinyl-N-methylphenothiazine) showed excellent cycling stability (after 10 000 cycles at a 10C rate, 93% of the initial capacity was retained) in addition to a high rate capability because of supramolecular hole transport. We propose this concept to be used in the future design of redox polymers for batteries.


 Please wait while we load your content...
Please wait while we load your content...