Linker-controlled polymeric photocatalyst for highly efficient hydrogen evolution from water†
Abstract
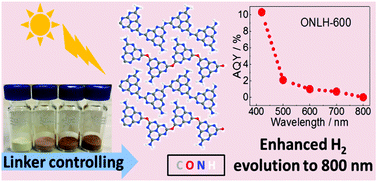
Polymeric photocatalysts have been identified as promising materials for H2 production from water due to their comparative low cost and facile modification of the electronic structure. However, the majority only respond to a limited wavelength region (λ < 460 nm) and exhibit fast charge recombination. Our density-functional theory (DFT) calculations have identified an oxygen-doped polymeric carbon nitride structure with heptazine chains linked both by oxygen atoms and by nitrogen species, which results in a reduced band gap and efficient charge separation. A novel synthetic method has then been developed to control both surface hydrophilicity and more importantly, the linker species in a polymer, which highly influences the band gap and charge separation. As such, the synthesized polymer can be excited from UV via visible to even near-IR (λ = 800 nm) wavelengths, resulting in a 25 times higher H2 evolution rate (HER) than the previous benchmark polymeric g-C3N4 (λ > 420 nm), with an apparent quantum yield (AQY) of 10.3% at 420 nm and 2.1% at 500 nm, measured under ambient conditions, which is closer to the real environment (instead of vacuum conditions). The strategy used here thus paves a new avenue to dramatically tune both the light absorption and charge separation to increase the activity of polymeric photocatalysts.


 Please wait while we load your content...
Please wait while we load your content...