The role of iodide in the formation of lithium hydroxide in lithium–oxygen batteries†
Abstract
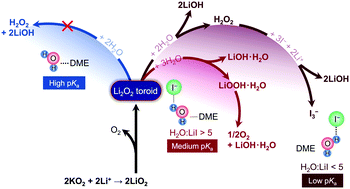
Lithium iodide has been studied extensively as a redox-mediator to reduce the charging overpotential of Li–oxygen (Li–O2) batteries. Ambiguities exist regarding the influence of lithium iodide on the reaction product chemistry and performance of lithium–oxygen batteries. In this work, we examined the role of lithium iodide on the reduction product chemistry under two conditions: (i) mixing KO2 with lithium salts and (ii) discharging Li–oxygen batteries at high and low overpotentials, in the presence of an ether-based electrolyte with different ratios of H2O : LiI. The addition of iodide to electrolytes containing water was found to promote the formation of LiOOH·H2O, LiOH·H2O and LiOH at the expense of Li2O2. At low H2O : LiI ratios (lower than 5), LiOH instead of Li2O2 was formed, which was accompanied by the oxidation of iodide to triodide while at high H2O : LiI ratios (12, 24, 134), a mixture of Li2O2, LiOOH·H2O and LiOH·H2O was observed and no triiodide was detected. The reaction between peroxide Li2O2 and/or superoxide LiO2 with H2O to form LiOH is facilitated by increased water acidity by strong I−–H2O interactions as revealed by 1H NMR and FT-IR measurements. This mechanism of LiOH formation in the presence of LiI and H2O was also found upon Li–O2 cell discharge, which is critical to consider when developing LiI as a redox mediator for Li–O2 batteries.


 Please wait while we load your content...
Please wait while we load your content...