Direct observation of enhanced water and carbon dioxide reactivity on multivalent metal oxides and their composites†
Abstract
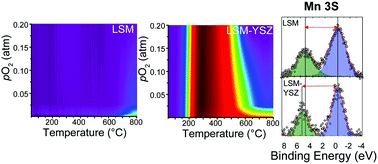
Electrochemical gas–solid reactions are important for many energy technologies, involving partial pressure dependent surface catalysis and bulk conduction phenomenon unresolvable using conventional techniques. We report for the first time water and CO2 reactivity on perovskites, fluorites, and their composites across a wide temperature- oxygen partial pressure (pO2) range by in situ gas phase isotopic oxygen exchange, demonstrating that multivalent metal oxides actively “breathe” oxygen from these gasses. We reveal the importance of cation oxidation state and the roles of electronic, ionic, and mixed conductor phases, as well as their composites. Moreover, we show that an ionic and electronic conducting oxide composite dramatically modifies the kinetics of oxygen exchange as compared to these materials individually or as compared to the effect of creating a composite from two mixed conducting oxides.


 Please wait while we load your content...
Please wait while we load your content...