Electrochemical reduction of CO2 to synthesis gas with controlled CO/H2 ratios†
Abstract
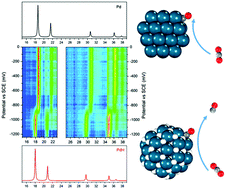
The electrochemical carbon dioxide reduction reaction (CO2RR) to simultaneously produce carbon monoxide (CO) and hydrogen (H2) has been achieved on carbon supported palladium (Pd/C) nanoparticles in an aqueous electrolyte. The synthesis gas product has a CO to H2 ratio between 0.5 and 1, which is in the desirable range for thermochemical synthesis of methanol and Fischer–Tropsch reactions using existing industrial processes. In situ X-ray absorption spectroscopy in both near-edge (XANES) and extended regions (EXAFS) and in situ X-ray diffraction show that Pd has transformed into β-phase palladium hydride (β-PdH) during the CO2RR. Density functional theory (DFT) calculations demonstrate that the binding energies of both adsorbed CO and H are significantly weakened on PdH than on Pd surfaces, and that these energies are potential descriptors to facilitate the search for more efficient electrocatalysts for syngas production through the CO2RR.


 Please wait while we load your content...
Please wait while we load your content...