Nanocrystalline LaOx/NiO composite as high performance electrodes for supercapacitors
Abstract
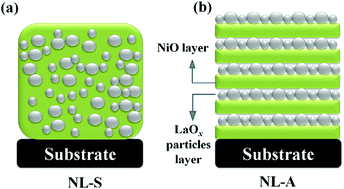
Nanocrystalline LaOx/NiO composite electrodes were synthesized via two types of facile cathodic electrodeposition methods onto nickel foam followed by thermal annealing without any binders. Scanning electron microscopy and transmission electron microscopy investigation revealed that LaOx nanocrystalline particles with an average diameter of 50 nm are uniformly distributed in the NiO layer or alternately deposited with the NiO layer onto the substrate. It is speculated that LaOx particles can participate in the faradaic reaction directly and offer more redox sites. Besides this, the unique Ni/La layered structure facilitates the diffusion of ions and retards the electrode polarization, thus leading to a better rate capability and cycling stability of NiO. As a result, the obtained electrodes display very competitive electrochemical performance (a specific capacitance of 1238 F g−1 at a current density of 0.5 A g−1, excellent rate capability of 86% of the original capacitance at 10 A g−1 and excellent cycling stability of 93% capacitance after 10 000 cycles). In addition, asymmetric coin devices were assembled using LaOx/NiO as the positive electrode and active carbon as the negative electrode. The assembled asymmetric devices demonstrate a high energy density of 13.12 W h kg−1 at a power density of 90.72 W kg−1.


 Please wait while we load your content...
Please wait while we load your content...