A series of Mg–Zn heterometallic coordination polymers: synthesis, characterization, and fluorescence sensing for Fe3+, CS2, and nitroaromatic compounds†
Abstract
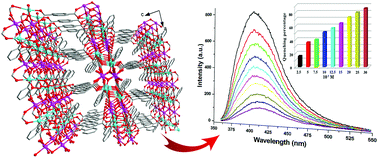
Herein, the solvothermal preparations, crystal structures, and fluorescence properties of three Mg–Zn heterometallic coordination polymers (CPs), i.e. [Mg2Zn2(OH)2(1,4-NDC)3(H2O)2]·6H2O (1), [Mg1.13Zn1.87(1,4-NDC)3(dppe)(CH3OH)] (2), and [Mg1.17Zn1.83(1,4-NDC)3(py)2]·1.5py (3), based on the mixed ligands of 1,4-naphthalene dicarboxylic acid (1,4-H2NDC) and N-containing ligands of 1,3-di(4-pyridyl)propane (dppe) or pyridine (py) are presented. In the title compounds, Zn and Mg are statistically distributed, as confirmed by single crystal analysis, and the ratios of Zn and Mg have been identified by inductively coupled plasma (ICP) measurements. In compound 1, the metal ions are tetra- or hexa-coordinated by the oxygen atoms of the carboxylate and μ3-OH group to form a one-dimensional (1D) ribbon, and these 1D ribbons are further bridged by 1,4-NDC linkers to grow into a 3D framework with 1D channels along the b axis. In compounds 2 and 3, the metal ions also adopt tetra- or hexa-coordination modes and are inter-bridged by carboxylate oxygen atoms to form a tri-nuclear secondary building unit (SBU). Due to the introduction of N-containing ligands acting as terminal molecules, these SBUs are linked by 1,4-NDC ligands to form a 2D network. Photoluminescence (PL) studies indicated that the title compounds showed strong blue emissions. Significantly, compound 1 demonstrated sensitive fluorescence sensing for Fe3+, carbon disulfide (CS2), and nitroaromatic compounds at low concentrations. The fluorescence sensing properties of compound 2 were also comparatively investigated in detail.


 Please wait while we load your content...
Please wait while we load your content...