Nanostructured N-doped orthorhombic Nb2O5 as an efficient stable photocatalyst for hydrogen generation under visible light†
Abstract
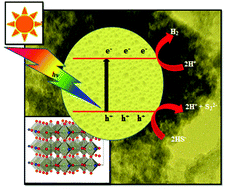
The synthesis of orthorhombic nitrogen-doped niobium oxide (Nb2O5−xNx) nanostructures was performed and a photocatalytic study carried out in their use in the conversion of toxic H2S and water into hydrogen under UV-Visible light. Nanostructured orthorhombic Nb2O5−xNx was synthesized by a simple solid-state combustion reaction (SSCR). The nanostructural features of Nb2O5−xNx were examined by FESEM and HRTEM, which showed they had a porous chain-like structure, with chains interlocked with each other and with nanoparticles sized less than 10 nm. Diffuse reflectance spectra depicted their extended absorbance in the visible region with a band gap of 2.4 eV. The substitution of nitrogen in place of oxygen atoms as well as Nb–N bond formation were confirmed by X-ray photoelectron spectroscopy (XPS) and Raman spectroscopy. A computational study (DFT) of Nb2O5−xNx was also performed for investigation and conformation of the crystal and electronic structure. N-Substitution clearly showed a narrowing of the band gap due to N 2p bands cascading above the O 2p band. Considering the band gap in the visible region, Nb2O5−xNx exhibited enhanced photocatalytic activity toward hydrogen evolution (3010 μmol h−1 g−1) for water splitting and (9358 μmol h−1 g−1) for H2S splitting under visible light. The enhanced photocatalytic activity of Nb2O5−xNx was attributed to its extended absorbance in the visible region due to its electronic structure being modified upon doping, which in turn generates more electron–hole pairs, which are responsible for higher H2 generation. More significantly, the mesoporous nanostructure accelerated the supression of electron and hole recombination, which also contributed to the enhancement of its activity.


 Please wait while we load your content...
Please wait while we load your content...