A cell-selective glutathione-responsive tris(phthalocyanine) as a smart photosensitiser for targeted photodynamic therapy†
Abstract
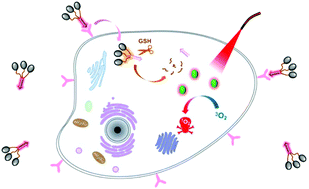
A biotin-conjugated disulfide-linked tris(phthalocyanine) has been synthesised and characterised. As shown by electronic absorption and steady-state fluorescence spectroscopy methods, the compound remains non-aggregated in N,N-dimethylformamide, but is significantly aggregated in phosphate buffered saline with 1% Cremophor EL. The reduction in fluorescence intensity and singlet oxygen generation efficiency as compared with the monomeric counterpart suggests that the tris(phthalocyanine) exhibits an excellent self-quenching effect, particularly in an aqueous medium. Upon interaction with glutathione, both the fluorescence emission and singlet oxygen production can be restored as a result of the cleavage of the disulfide linkages, thereby releasing the phthalocyanine units and reducing their aggregation and self-quenching effects. With a biotin moiety, this tris(phthalocyanine) is preferentially taken up by the biotin-receptor-positive HeLa cells and activated by the intracellular glutathione, resulting in fluorescence recovery and photocytotoxicity with an IC50 value of 0.68 μM. This compound can therefore act as a dual functional photosensitiser.


 Please wait while we load your content...
Please wait while we load your content...