In vitro/in vivo “peeling” of multilayered aminocarboxylate gold nanoparticles evidenced by a kinetically stable 99mTc-label†
Abstract
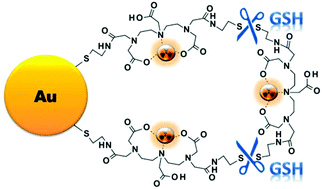
A thiolated bombesin peptide was conjugated to Au-DTDTPA nanoconstructs to obtain BBN-Au-DTDTPA targeted to the gastrin releasing peptide receptor (GRPr). Different analytical techniques showed that this conjugate shares similar physico-chemical properties with Au-DTDTPA; HPLC and XPS analyses corroborated the attachment of the bioactive peptide to the AuNPs surface. Competitive binding assays in PC3 cancer cells showed that these BBN-containing AuNPs have high affinity for GRPr. BBN-Au-DTDTPA was successfully radiolabeled with 99mTc and showed high in vitro stability towards different biological media and substrates, except for glutathione (GSH). In vitro and in vivo studies, based on gamma-counting (99mTc content) and neutron activation analysis (Au content), indicated the release of the DTDTPA coating from the AuNPs. Probably, the “peeling” of the layered-aminocarboxylate coating is GSH-mediated and involves the cleavage of the DTDTPA disulfide bonds and/or Au–S bonds. These results render BBN-Au-DTDTPA an interesting platform deserving further evaluation in target-specific GSH-mediated drug delivery.
- This article is part of the themed collection: Frontiers in Radionuclide Imaging and Therapy


 Please wait while we load your content...
Please wait while we load your content...