Rescaling metal molybdate nanostructures with biopolymer for energy storage having high capacitance with robust cycle stability
Abstract
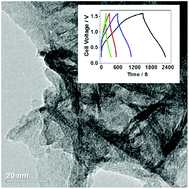
Hybrid capacitors can replace or complement batteries, while storing energy through ion adsorption and fast surface redox reactions. There is a growing demand in developing nanostructured materials as electrodes for hybrid systems that can enhance the specific capacitance by ion desolvation in the nanopores. Here, we demonstrate that rescaling the pore diameter with the aid of biopolymer at an optimal level during the synthesis of metal molybdate leads to high capacitance 124 F g−1 giving robust capacitance retention of 80% over 2000 cycles for a constructed device (activated carbon vs. metal molybdate). The presence of biopolymer (L-glutamic acid) in the metal molybdate acts as a complexing agent of the metal ion while enhancing the mass transport and hence it's improved electrochemical performance. However, XPS and other elemental analyses illustrated no evidence for N doping but traces of other surface functional groups (i.e. C and O) could be present on the molybdate surface. The biopolymer synthetic approach has the advantage of yielding nanostructured material with a relatively narrow pore size distribution controlled by L-glutamic acid. This study will provide a generic route to rescale other metal molybdate, phosphate or oxide counterparts and be an added value to the database.


 Please wait while we load your content...
Please wait while we load your content...