Ditopic receptors containing urea groups for solvent extraction of Cu(ii) salts†
Abstract
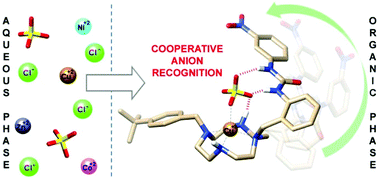
The ditopic receptor L3 [1-(2-((7-(4-(tert-butyl)benzyl)-1,4,7,10-tetraazacyclododecan-1-yl)methyl)phenyl)-3-(3-nitrophenyl)urea] containing a macrocyclic cyclen unit for Cu(II)-coordination and a urea moiety for anion binding was designed for recognition of metal salts. The X-ray structure of [CuL3(SO4)] shows that the sulfate anion is involved in cooperative binding via coordination to the metal ion and hydrogen-bonding to the urea unit. This behaviour is similar to that observed for the related receptor L1 [1-(2-((bis(pyridin-2-ylmethyl)amino)methyl)phenyl)-3-(3-nitrophenyl)urea], which forms a dimeric [CuL1(μ-SO4)]2 structure in the solid state. In contrast, the single crystal X-ray structure of [ZnL3(NO3)2] contains a 1 : 2 complex (metal : anion) where one anion coordinates to the metal and the other is hydrogen-bonded to the urea group. Spectrophotometric titrations performed for the [CuL3(OSMe2)]2+ complex indicate that this system is able to bind a wide range of anions with an affinity sequence: MeCO2− > Cl− > H2PO4− > Br− > NO2− > HSO4− > NO3−. Lipophilic analogues of L1 and L3 extract CuSO4 and CuCl2 from water into chloroform with high selectivity over the corresponding Co(II), Ni(II) and Zn(II) salts.


 Please wait while we load your content...
Please wait while we load your content...