A p-nitroaniline redox-active solid-state electrolyte for battery-like electrochemical capacitive energy storage combined with an asymmetric supercapacitor based on metal oxide functionalized β-polytype porous silicon carbide electrodes†
Abstract
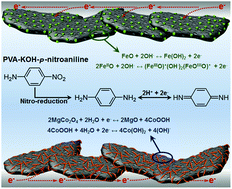
A unique redox active flexible solid-state asymmetric supercapacitor with ultra-high capacitance and energy density was fabricated using a composite comprising MgCo2O4 nanoneedles and micro and mesoporous silicon carbide flakes (SiCF) (SiCF/MgCo2O4) as the positive electrode material. Due to the synergistic effect of the two materials, this hybrid electrode has a high specific capacitance of 516.7 F g−1 at a scan rate of 5 mV s−1 in a 1 M KOH aqueous electrolyte. To obtain a reasonable matching of positive and negative electrode pairs, a composite of Fe3O4 nanoparticles and SiCF (SiCF/Fe3O4) was synthesized for use as a negative electrode material, which shows a high capacitance of 423.2 F g−1 at a scan rate of 5 mV s−1. Therefore, by pairing the SiCF/MgCo2O4 positive electrode and the SiCF/Fe3O4 negative electrode with a redox active quasi-solid-state PVA-KOH-p-nitroaniline (PVA-KOH-PNA) gel electrolyte, a novel solid-state asymmetric supercapacitor device was assembled. Because of the synergistic effect between the highly porous SiCF and the vigorous redox-reaction of metal oxides, the hybrid nanostructure electrodes exhibited outstanding charge storage and transport. In addition, the redox active PVA-KOH-PNA electrolyte adds additional pseudocapacitance, which arises from the nitro-reduction and oxidation and reduction process of the reduction product of p-phenylenediamine, resulting in an enhancement of the capacitance (a specific capacitance of 161.77 F g−1 at a scan rate of 5 mV s−1) and energy density (maximum energy density of 72.79 Wh kg−1 at a power density of 727.96 W kg−1).


 Please wait while we load your content...
Please wait while we load your content...