Synthesis, characterization and magnetic studies on mono-, di-, and tri-nuclear Cu(ii) complexes of a new versatile diazine ligand†
Abstract
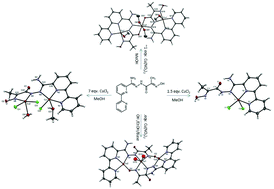
The synthesis and coordination chemistry of a new open-chain diazine ligand (L4H2) containing bipyridine, oxime and hydrazone functionalities is reported. Reaction of L4H2 with CuCl2·2H2O affords the mononuclear complex [Cu(L4H2)Cl2] (1) in which the ligand acts as a neutral tridentate N,N′,N′′ donor, whereas treatment with a large excess of CuCl2·2H2O affords the dinuclear complex [Cu2(L4H)Cl3(CH3OH)(H2O)] (2) in which the ligand is singly-deprotonated at the diazine, offering N,N′,N′′ and N,N′ donor sets to two Cu(II) ions. Reaction with Cu(NO3)2·3H2O yields the trinuclear complex, [Cu3(L4H)2(CH3OH)4(NO3)4] (3) in which the ligand is again singly deprotonated, but now presents a tetradentate N,N′,N′′,N′′′-donor set to the first Cu(II) and behaves as an N,O-chelate to a second Cu(II), with a conformational change from cis to trans at the diazine moiety. When the latter reaction is repeated in the presence of a mild base, a second trinuclear complex is isolated, [Cu3(L4)2(CH3CH2OH)](NO3)2 (4) in which both the diazine and oxime functionalities of the ligand are deprotonated which subsequently bridges all three Cu(II) ions, acting as an N,N′,N′′ donor to the first Cu(II), a N,N′ donor to a second Cu(II) (similar to 2) and as a terminal O-donor to a third Cu(II) ion. The Cu(II) ions are linked mutually cis via the two-atom diazine bridge in the case of 2 and 4 and trans in the case of 3. Magnetic studies reveal the presence of weak ferromagnetic interactions in 2 (g = 2.2, J/k = +12.8 K) and strong antiferromagnetic interactions in both 3 and 4 (g = 2.093 J/k = −140 K and g = 2.24 J/k = −300 K respectively).


 Please wait while we load your content...
Please wait while we load your content...