Heterodinuclear titanium/zinc catalysis: synthesis, characterization and activity for CO2/epoxide copolymerization and cyclic ester polymerization†
Abstract
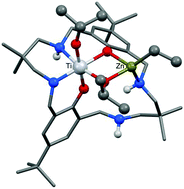
The preparation of heterodinuclear complexes, especially those comprising early-late transition metals coordinated by a simple or symmetrical ancillary ligand, represents a fundamental challenge and an opportunity to prepare catalysts benefitting from synergic properties. Here, two new mixed titanium(IV)–zinc(II) complexes, [LTi(OiPr)2ZnEt] and [LTi(OiPr)2ZnPh], both coordinated by a diphenolate tetra(amine) macrocyclic ligand (L), are prepared. The synthesis benefits from the discovery that reaction of the ligand with a single equivalent of titanium tetrakis(iso-propoxide) allows the efficient formation of a mono-Ti(IV) complex, [LTi(OiPr)2]. All new complexes are characterized by a combination of single crystal X-ray diffraction, multinuclear NMR spectroscopy and mass spectrometry techniques. The two heterobimetallic complexes, [LTi(OiPr)2ZnEt] and [LTi(OiPr)2ZnPh], feature trianionic coordination by the macrocyclic ligand and bridging alkoxide groups coordinate to both the different metal centres. The heterodinuclear catalysts are compared to the mono-titanium analogue, [LTi(OiPr)2], in various polymerization reactions. In the alternating copolymerizations of carbon dioxide and cyclohexene oxide, the mono-titanium complex is totally inactive whilst the heterodinuclear complexes show moderate activity (TOF = 3 h−1); it should be noted the activity is measured using just 1 bar pressure of carbon dioxide. In the ring opening polymerization of lactide and ε-caprolactone, the mono-Ti(IV) complex is totally inactive whilst the heterodinuclear complexes show moderate-high activities, qualified by comparison to other known titanium polymerization catalysts (L-lactide, kobs = 11 × 10−4 s−1 at 70 °C, 1 M in [lactide]) and ε-caprolactone (kobs = 5 × 10−4 s−1 at 70 °C, 0.9 M in [ε-caprolactone]).


 Please wait while we load your content...
Please wait while we load your content...