Photocatalytic reduction of CO2 with H2O vapor under visible light over Ce doped ZnFe2O4
Abstract
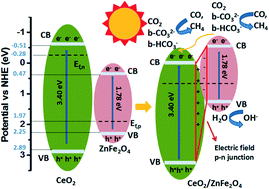
In this work, ZnFe2O4 spinels doped with different Ce contents were synthesized through a sol–gel method and their morphologies, structures, optical and electronic properties, adsorption capacity for CO2 and activities for CO2 photoreduction with H2O vapor were investigated. The results show that the presence of Ce3+ in the ZnFe2O4 lattice and CeO2 particles on the surface of ZnFe2O4 can enhance photoabsorption in the visible light region, and promote the effective separation of photogenerated electron–hole pairs. CO2-TPD and in situ FTIR spectroscopy confirmed that monodentate carbonate (m-CO32−), bidentate carbonate (b-CO32−) and bidentate bicarbonate (b-HCO3−) were the main surface species for the coadsorption of CO2 and H2O on photocatalyst surfaces. The addition of Ce strengthens the chemical interaction between CO2 and photocatalyst surfaces and obviously increases the adsorption amount of b-HCO3− and b-CO32− species. As a result, all Ce-doped samples exhibit much higher production rates of H2, CO and CH4 when compared to undoped ZnFe2O4, and ZFC0.2 has the highest photocatalytic activity among all samples, for which H2, CO and CH4 production rates of 19.9, 1.7 and 9.9 μmol gcat−1 h−1 were achieved, about 4.6, 4.3 and 4.2 times that of undoped ZnFe2O4, respectively.


 Please wait while we load your content...
Please wait while we load your content...