Insight into the effect of incorporation of boron into ZSM-11 on its catalytic performance for conversion of methanol to olefins†
Abstract
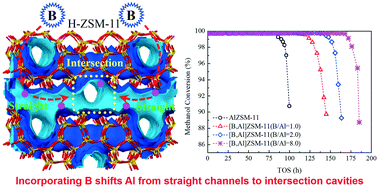
A series of ZSM-11 zeolites was synthesized by adding different amounts of boron to a synthesis gel for the purpose of improving its catalytic performance for conversion of methanol to olefins (MTO). It was found that boron has marginal effects on the crystal structure, texture properties and acid site strength of ZSM-11, but it significantly increases its catalytic stability and hydride transfer activity that results in an increase in the amounts of aromatics and alkanes in products. DFT calculations revealed that aluminum and boron atoms competitively enter the ZSM-11 framework. In combination with 27Al MAS NMR, GC-MS, m-xylene isomerization and Co2+-exchanged ZSM-11 diffuse reflectance UV-vis spectroscopy results, it can be deduced that incorporation of boron compels some aluminum atoms to shift towards the intersection T sites, which can greatly alleviate the coking process although it allows the formation of more bulky molecules. This shows that the MTO catalytic performance of ZSM-11 can be largely adjusted by changing the amount of incorporated boron through regulation of aluminum or acid site locations in the framework.


 Please wait while we load your content...
Please wait while we load your content...