Selective hydrodeoxygenation of tartaric acid to succinic acid†
Abstract
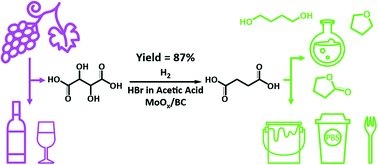
A novel one-step process for catalytic production of succinic acid from tartaric acid, which is largely available in the waste streams of wine making, is developed. A liquid-phase system comprised of a molybdenum oxide catalyst supported on carbon black (MoOx/BC) and hydrobromic acid in acetic acid under a H2 atmosphere is effective for selective cleavage of the C–O bonds of tartaric acid and selective hydrogenation of the resulting unsaturated carbons. Temperature, hydrogen pressure, and catalyst pre-treatment are optimized to understand their impact on catalytic activity and product distribution, leading to an 87% yield of succinic acid at 170 °C. Importantly, reduction of the carboxyl groups and C–C bond cleavage are retarded. Pre-reduction and characterization studies (TPR, XRD, XPS, and XAS) reveal that the high catalyst activity of MoOx/BC is correlated with the lower Mo oxidation states (+4 to 0) formed during pre-reduction that promote cleavage of the C–O bonds of tartaric acid and hydrogenation of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond of the intermediate fumaric acid. Recyclability studies and structural characterization of the recovered catalyst indicate that MoOx/BC remains active upon reuse.
C bond of the intermediate fumaric acid. Recyclability studies and structural characterization of the recovered catalyst indicate that MoOx/BC remains active upon reuse.


 Please wait while we load your content...
Please wait while we load your content...