Amine coupled ordered mesoporous (Co–N) co-doped TiO2: a green photocatalyst for the selective aerobic oxidation of thioether†
Abstract
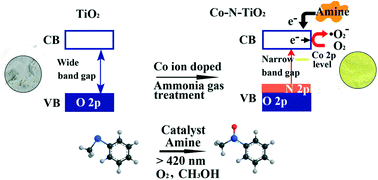
Usually oxygen cannot participate in the selective oxidation reaction of thioether to sulfoxide, which is attributed to its inactive feature. Although UV-light irradiation can activate O2, this process also results in UV-induced hole (hvb+) generation. This oxidative species and O2 can be consumed for the formation of free radical intermediates that leads to uncontrolled auto-oxidation products resulting in low selectivity. Current work aims to develop an approach for the aerobic oxidation of thioether to sulfoxide by exploring the synergy between triethylamine and mesoporous Co–N–TiO2 (nitrided @ 500 °C for 2 h) microspheres (with a large surface area: 124 m2 g−1); the oxidation is carried out via visible-light photoredox catalysis. Triethylamine, due to its electron donating ability, acts as a redox mediator. With a lower band gap and level of conduction band minimum (−0.605 V vs. NHE), the Co–N–TiO2 sample has the ability to be an efficient and selective catalyst. Excellent conversion from the selective oxidation of thioanisole (76.4%) under 12 h visible light irradiation is observed. It may be noted that Co doping into TiO2 alone does not aid in obtaining a good photocatalyst; in fact this sample shows the Moss–Burstein effect. We have also shown that the reported photocatalyst is applicable for the conversion of several other thioethers to sulfoxides; generally high conversion rates and selectivities are observed. The reaction mechanisms are studied using UV-visible absorption spectra and the oxidation–extraction photometry (OEP) method. This result is likely useful for further exploration of surface complex photocatalysts for many other aerobic oxidation reactions.


 Please wait while we load your content...
Please wait while we load your content...