Transient structural and catalytic behaviour of Pt-particles probed by operando spectroscopy during a realistic driving cycle†
Abstract
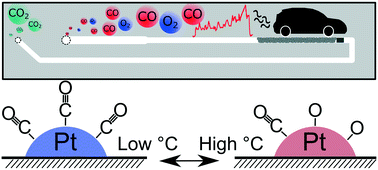
Pt-based diesel oxidation catalysts were investigated for CO oxidation activity under rapid transient temperature conditions based on a realistic driving cycle, which is presently a focal point in exhaust gas aftertreatment. Experiments were performed in a microreactor setup allowing rapid heating/cooling coupled with operando Turbo X-ray absorption spectroscopy (T-XAS) and on-line product analysis by mass spectrometry. Significant differences were observed in catalyst structure and performance depending on the temperature ramp rate. Particularly for Pt/Al2O3, the Pt oxidation state followed a dynamic hysteresis profile during CO oxidation light-off and light-out. In contrast, in Pt–CeO2/Al2O3, ceria acted as an oxygen storage buffer, reducing the width of the Pt oxidation/reduction hysteresis loop as a function of the temperature ramp rate. Ceria also supplied oxygen to the Pt surface, helping to maintain high activity during cooling down and at lower temperatures during transient conditions. This study shows the potential insights into the reaction mechanism available when considering transient temperature as an experimental condition during operando spectroscopic studies in exhaust gas catalysis. The current method is applicable to virtually any rapid transient temperature driving cycle.
- This article is part of the themed collection: 2017 Catalysis Science & Technology HOT Articles


 Please wait while we load your content...
Please wait while we load your content...