Palladium–gold single atom alloy catalysts for liquid phase selective hydrogenation of 1-hexyne†
Abstract
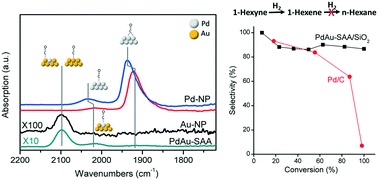
Silica supported and unsupported PdAu single atom alloys (SAAs) were investigated for the selective hydrogenation of 1-hexyne to hexenes under mild conditions. The catalysts were prepared by adding a trace amount of Pd (0.4 at%) into the surface of pre-formed Au nanoparticles through a sequential reduction method. TEM and XRD analyses indicate the formation of PdAu nanoparticles and ATR-IR confirms the single atom dispersion of Pd in the Au matrix. In time-resolved batch reactor studies, we found that the Pd single atoms improved the hydrogenation activity of Au by nearly 10-fold but did not decrease the high selectivity to partial hydrogenation products. The enhanced reactivity is attributed to the Pd single atoms (isolated Pd atoms in the Au surface) facilitating molecular hydrogen dissociation leading to the availability of weakly bound atomic hydrogen on the otherwise inert gold surface. Higher than 85% selectivity to hexenes was observed, which is significantly greater than that of monometallic Pd catalysts. Model catalyst studies were conducted to investigate the formation and reactivity of the Pd/Au(111) SAAs. Scanning tunneling microscopy of Pd/Au(111) surfaces confirms the formation of PdAu single atom alloys at low Pd coverage with the Pd preferentially located in the vicinity of the herringbone elbows of the reconstructed Au(111) surface. Temperature-programmed desorption experiments confirm that single Pd atom sites dissociate hydrogen and bind both CO and H atoms more weakly as compared to extended Pd surfaces.
- This article is part of the themed collection: Single atom catalysis


 Please wait while we load your content...
Please wait while we load your content...