Minireview: direct catalytic conversion of sour natural gas (CH4 + H2S + CO2) components to high value chemicals and fuels
Abstract
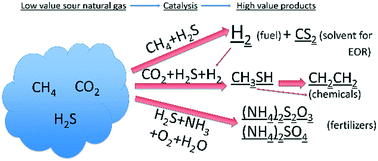
The abundance of natural gas reserves can facilitate the transition from fossil derived to fully renewable chemical and fuel generation. Natural gas, however, can contain more than 10% sour gas molecules (chiefly CO2 and H2S), especially in the Middle East and Russia. Currently, the most common sour natural gas separation technologies rely on the use of various amine-based absorption–desorption cycles in absorption towers, membrane separation and cryogenic processes. However, these processes are quite expensive and, unfortunately, result in the necessity for further processing of the sour gas stream. We feel that the implementation of catalytic activation of the sour gas stream, so as to form useful products, has been underexplored and thus has yet to be fully developed. In this paper, we present a series of catalytic reactions for the sour gas components so as to obtain useful chemicals or fuels, such as carbon disulfide (CS2), hydrogen (H2), ethylene (C2H4), ammonium sulfate ((NH4)2SO4) and ammonium thiosulfate ((NH4)2S2O3). Particular catalytic reactions which can form those products are (a) CH4 reforming with H2S on supported metal oxide catalysts, (b) CO2 + H2S + H2 catalytic reaction on K2O/WO3/Al2O3 to yield a CH3SH intermediate, (c) olefin production via CH3SH coupling on zeolites and supported metal oxides and (d) H2S + NH3 catalytic reaction to yield ammonium sulfate ((NH4)2SO4) and ammonium thiosulfate ((NH4)2S2O3) on partial oxidation catalysts. Thermodynamic considerations, together with catalyst design techniques, are discussed for each reaction.
- This article is part of the themed collection: 2017 Catalysis Science & Technology HOT Articles


 Please wait while we load your content...
Please wait while we load your content...