Iridium–NSiN catalyzed formation of silylphosphinecarboxylates from the reaction of CO2 with P(SiMe3)R2 (R = Ph, Cy)†
Abstract
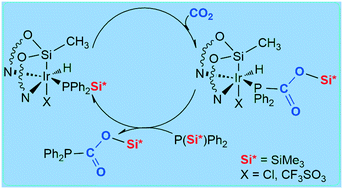
The iridium–NSiN species [Ir(H)(X)(NSiN)(coe)] (coe = cis-cyclooctene; NSiN = bis(pyridine-2-yloxy)methylsilyl fac-coordinated; X = Cl, (1); CF3SO3, (2)) have been proven to be effective catalyst precursors for the formation of silylphosphinecarboxylates (P{CO2SiMe3}(R)2 (R = Ph, (3a); Cy (3b))) by the reaction of CO2 with the corresponding silyl phosphine P(SiMe3)R2 (R = Ph, Cy) under mild conditions. The ancillary ligand at the Ir–NSiN active species plays a role in the reaction mechanism. Thus, complex 2 with a triflate ligand has been found to be more active than the chloride derivative 1. The proposed mechanism based on stoichiometric experiments and theoretical calculations at the DFT level shows that the ancillary ligand assisted P–Si bond activation process could be considered the rate-determining step of the overall catalytic cycle.


 Please wait while we load your content...
Please wait while we load your content...