Transition-metal-free B–B and B–interelement reactions with organic molecules
Abstract
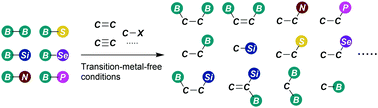
This review is a guided tour along the activation modes and reactivity of B–B, B–Si, B–N, B–S, B–Se and B–P reagents, in the absence of any transition metal complex. Here are disclosed the general concepts related to the homolytic and heterolytic cleavage of B–B and B–interelement bonds, as well as the generation of new C–B and C–interelement bonds, in a selective way. The greener consequences of these novel routes facilitate the gram scale preparations of target functionalised organic compounds. Intrinsic data about the suggested mechanisms and spectroscopic evidence that supports the innovative theories are provided along the review. Since this is a stimulating area of work that has emerged within the last decade, this overview serves as the basis to understand the new trends and hopefully to generate inspiration for future discoveries in the field.

 Please wait while we load your content...
Please wait while we load your content...