Retracted Article: The reductive phase of Rhodobacter sphaeroides cytochrome c oxidase disentangled by CO ligation†
Abstract
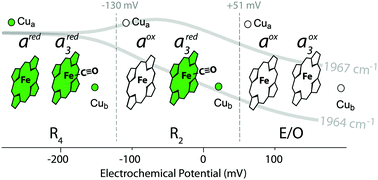
Cytochrome c oxidase (CcO) is a membrane protein of the respiratory chain that catalytically reduces molecular oxygen (O2) to water while translocating protons across the membrane. The enzyme hosts two copper and two heme iron moieties (heme a/heme a3). The atomic details of the sequential steps that go along with this redox-driven proton translocation are a matter of debate. Particularly for the reductive phase of CcO that precedes oxygen binding experimental data are scarce. Here, we use CcO under anaerobic conditions where carbon monoxide (CO) is bound to heme a3 which in tandem with CuB forms the binuclear center (BNC). Fourier-transform infrared (FTIR) absorption spectroscopy is combined with electro-chemistry to probe different redox and protonation states populated by variation of the external electrostatic potential. With this approach, the redox behavior of heme a and the BNC could be separated and the corresponding redox potentials were determined. We also infer the protonation of one of the propionate side chains of heme a3 to correlate with the oxidation of heme a. Experimental changes in the local electric field surrounding CO bound to heme a3 are determined by their vibrational Stark effect and agree well with electrostatic computations. The comparison of experimental and computational results indicates that changes of the heme a3/CuB redox state are coupled to proton transfer towards heme a3. The latter supports the role of the heme a3 propionate D as proton loading site.


 Please wait while we load your content...
Please wait while we load your content...