Vibrational states of nano-confined water molecules in beryl investigated by first-principles calculations and optical experiments†
Abstract
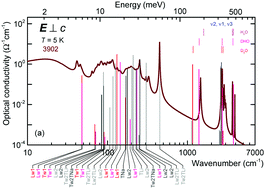
Using quantum mechanical calculations within density functional theory, we provide a comprehensive analysis of infrared-active excitation of water molecules confined in nanocages of a beryl crystal lattice. We calculate infrared-active modes including the translational, librational, and mixed-type resonances of regular and heavy water molecules. The results are compared to the experimental spectra measured for the two principal polarizations of the electric field: parallel and perpendicular to the crystallographic c-axis. Good agreement is achieved between calculated and measured isotopic shifts of the normal modes. We analyze the vibrational modes in connection with the structural characteristics and arrangements of water molecules within the beryl crystal. Specific atomic displacements are assigned to each experimentally detected vibrational mode resolving the properties of nano-confined water on scales not accessible by experiments. Our results elucidate the applicability and efficiency of a combined experimental and computational approach for describing and an in-depth understanding of nano-confined water, and pave the way for future studies of more complex systems.


 Please wait while we load your content...
Please wait while we load your content...