Exploring the solid state phase transition in dl-norvaline with terahertz spectroscopy†
Abstract
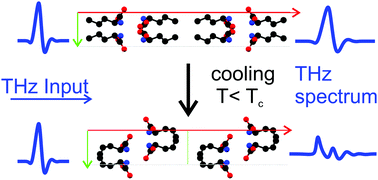
DL-Norvaline is a molecular crystal at room temperature and it undergoes a phase transition when cooled below 190 K. This phase transition is believed to be Martensitic, thus making it of particular interest for molecular machines. In this paper we investigate this phase transition by measuring its terahertz (THz) spectrum over a range of temperatures. Temperature-dependent THz time-domain spectroscopy (THz-TDS) measurements reveal that the transition temperature (Tβ→α) is 190 K. The influence of nucleation seeds was analyzed by determining the Tβ→α of molecular crystals with varying grain size. Grains of 5 μm or less result in a lower transition temperature (Tβ→α = 180 K) compared to larger grains of 125–250 μm (Tβ→α = 190 K). Additionally, we gain insight into the physical process of the phase transition via temperature-dependent THz-TDS spectra of doped and mixed molecular crystals. The addition of molecular dopants, which differ from DL-norvaline only at the end of the side chain which resides in the hydrophobic layers of the crystal, decreases Tβ→α. This is consistent with a solid–solid phase transition in which the unit cell shifts along this hydrophobic layer, and it leads us to believe that the phase transition in DL-norvaline is Martensitic in nature.


 Please wait while we load your content...
Please wait while we load your content...