Effects of phosphate on biotite dissolution and secondary precipitation under conditions relevant to engineered subsurface processes†
Abstract
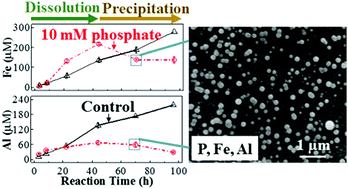
Brine–mica interfacial interactions affect both the caprock integrity and the fate and transport of reactive fluids at deep subsurface sites. Phosphate naturally exists at low concentration in subsurface brines, and its concentration can be increased significantly during energy-related engineered subsurface processes. However, our understanding of the influence of phosphate on brine–mica interactions is limited, especially under subsurface conditions. Here, biotite dissolution experiments were conducted without and with phosphate (0.1, 1, and 10 mM) at 95 °C and 102 atm CO2. Compared to the control, 0.1 mM, and 1 mM phosphate systems, biotite dissolution was four times higher with 10 mM phosphate. Despite the dissolution differences, in all the phosphate systems, phosphate interacted with Al and Fe sites in biotite, forming surface complexation and precipitating as Fe- or Al-bearing minerals on surfaces and in solutions. Consequently, aqueous Fe and Al concentrations became lower with phosphate than in the control experiments. In addition, the biotite basal surfaces became more hydrophilic after reaction with phosphate, even at 0.1 mM, mainly from phosphate adsorption. This study offers new information on how phosphate-containing brine interacts with caprocks and on the consequent wettability changes, results that can benefit current and future energy-related subsurface engineering processes.


 Please wait while we load your content...
Please wait while we load your content...