Clarifying the dehydrogenation pathway of catalysed Li4(NH2)3BH4–LiH composites†
Abstract
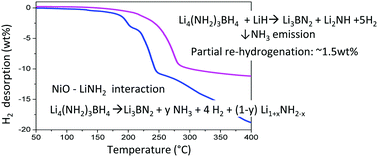
The effect of different metal oxides (Co3O4 and NiO) on the dehydrogenation reaction pathways of the Li4(NH2)3BH4–LiH composite was investigated. The additives were reduced to metallic species i.e. Co and Ni which act as catalysts by breaking the B–H bonds in the Li–B–N–H compounds. The onset decomposition temperature was lowered by 32 °C for the Ni-catalysed sample, which released 8.8 wt% hydrogen below 275 °C. It was demonstrated that the decomposition of the doped composite followed a mechanism via LiNH2 and Li3BN2 formation as the end product with a strong reduction of NH3 emission. The sample could be partially re-hydrogenated (∼1.5 wt%) due to lithium imide/amide transformation. To understand the role of LiH, Li4(NH2)3BH4–LiH–NiO and Li4(NH2)3BH4–NiO composites were compared. The absence of LiH as a reactant forced the system to follow another path, which involved the formation of an intermediate phase of composition Li3BN2H2 at the early stages of dehydrogenation and the end products LiNH2 and monoclinic Li3BN2. We provided evidence for the interaction between NiO and LiNH2 during heating and proposed that the presence of Li facilitates a NHx-rich environment and the Ni catalyst mediates the electron transfer to promote NHx coupling.


 Please wait while we load your content...
Please wait while we load your content...