Modelling pH and potential in dynamic structures of the water/Pt(111) interface on the atomic scale†
Abstract
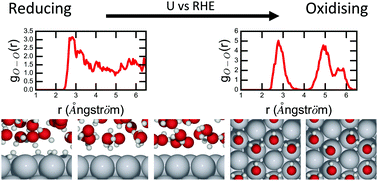
We present atomic-scale structures of the Pt(111)/water interface, by calculating distributions of atomic distances as functions of pH. The structure of the Pt(111)/water interface is a particularly interesting model system in electro-catalysis for proton exchange reactions, especially the oxygen reduction reaction in polymer electrolyte membrane fuel cells. Further insight into such reactions requires accurate simulations of the electrolyte structure in the interface. The study displays many interesting details in the behaviour of the electrolyte structure, e.g. that the electrolyte structure average responds to the presence of protons by a H-down water orientation and that hexagonal adsorbed water layers are present only when they are anchored at the surface by HO*. New adsorbate configurations were also found at 5/12 ML coverage of HO*, suggesting an explanation for reported cyclic voltammetry experiments. The present study is a step towards a more complete understanding of the structure of the electrochemical interface on the atomic scale.


 Please wait while we load your content...
Please wait while we load your content...