Covalent-reaction-induced interfacial assembly to transform doxorubicin into nanophotomedicine with highly enhanced anticancer efficiency†
Abstract
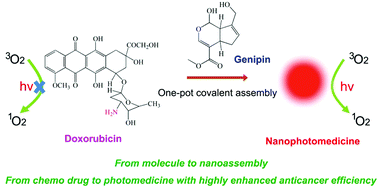
Herein, we show that a molecular assembly offers tremendous opportunities of affording existing building units with new physicochemical properties, holding promise in wide applications. Herein, we develop a facile covalent assembly using a natural occurring linker, genipin, to efficiently transform a traditional chemo drug, doxorubicin, into a nanophotomedicine. A possible mechanism is proposed, in which doxorubicin reacts with genipin through covalent bonding to produce poorly soluble units, which further form nuclei and mediate the interfacial assembly to generate uniform nanoparticles. Such assembled nanophotomedicine shows remarkably enhanced singlet oxygen generation ability (about 1000 folds), leading to a much higher photodynamic activity. Moreover, this self-carried nanodrug exhibits adjustable size, excellent colloidal stability, high capacity and preferable endocytosis. These favorable features lead to greatly improved anticancer efficiency under light at the same dosage, compared to that of pure doxorubin. We believe this study brings a new dimension to develop advanced drug delivery systems by molecular assembly.
- This article is part of the themed collections: 2017 PCCP HOT Articles and Surface chemistry and interface science


 Please wait while we load your content...
Please wait while we load your content...