Stability and growth mechanism of self-assembling putative antifreeze cyclic peptides†
Abstract
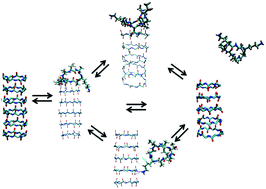
Cyclic peptides (CPs) that self-assemble in nanotubes can be candidates for use as antifreeze proteins. Based on the cyclic peptide sequence cyclo-[(L-LYS-D-ALA-L-LEU-D-ALA)2], which can stack into nanotubes, we propose a putative antifreeze cyclic peptide (AFCP) sequence, cyclo-[(L-LYS-D-ALA)2-(L-THR-D-ALA)2], containing THR-ALA-THR ice binding motifs. Using molecular dynamics simulations we investigate the stability of these cyclic peptides and their growth mechanism. Both nanotube sequences get more stable as a function of size. The relative stability of the AFCP sequence CPNT increases at sizes greater than a dimer by forming intermolecular THR side chain H-bonds. We find that, like the naturally occurring AF protein from spruce budworm (Choristoneura fumiferana), the THR distances of the AFCP's ice binding motif match the ice prism plane O–O distances, thus making the AFCP a suitable AF candidate. In addition, we investigated the nanotube growth process, i.e. the association/dissociation of a single CP to an existing AFCP nanotube, by Transition Path Sampling. We found a general dock-lock mechanism, in which a single CP first docks loosely before locking into place. Moreover, we identified several qualitatively different mechanisms for association, involving different metastable intermediates, including a state in which the peptide was misfolded inside the hydrophobic core of the tube. Finally, we find evidence for a mechanism involving non-specific association followed by 1D diffusion. Under most conditions, this will be the dominant pathway. The results yield insights into the mechanisms of peptide assembly, and might lead to an improved design of self-assembling antifreeze proteins.


 Please wait while we load your content...
Please wait while we load your content...